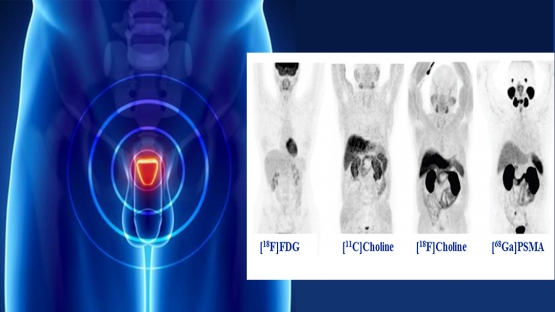
This Coordinated Research Project is the result of recommendations from the scientific community through interactions with IAEA technical officers. Prostate cancer is the most common solid malignancy in men and the third leading cause of cancer related morbidity and mortality in many Member States. In this context, the availability of a diagnostic test potentially able to differentiate between: 1. initial relapse, 2. loco-regional involvement, or 3. systemic recurrence, is of pivotal importance since the management approach will vary among these three clinical conditions. Imaging modalities are important for the definition of an optimal therapeutic approach and can guide treatment planning.
A new molecule was recently made available that targets the Prostate-Specific Membrane Antigen (PSMA) and can be labelled with positron emission radionuclides, such as Gallium 68, to perform PET/CT studies. The available scientific data show, in preliminary studies, that Ga 68 studies have a significantly higher accuracy than the currently use PET/CT with 18F-choline.
CRP overall objective:
The aim of this study is to evaluate the accuracy of 68Ga-PSMA PET/CT in detecting the presence of local and/or systemic disease in patients with prostate cancer.
Specific objectives will include:
1. To evaluate the performance of 68Ga-PSMA PET/CT compared to clinical disease prognostic factors (for example: the value of PSA at the time of PET, the PSADT, the PSA velocity (PSAvel), GS, TNM, the time between primary therapy and BR (TTR), ADT in place and age);
2. To evaluate the performance of 68Ga-PSMA PET/CT compared to the results of conventional imaging methods, used in the normal care pathway;
2. To evaluate the sensitivity, specificity and positive predictive value (PPV).
Many proposals were received for this CRP and 19 research institutes have already been awarded research contracts and agreements, therefore this CRP is no longer open for proposals. For further information related to this CRP, please contact Enrique Estrada-Lobato, Project Officer, Nuclear Medicine and Diagnostic Imaging Section, Division of Human Health, IAEA Department of Nuclear Science and Applications.